
Mycoplasma Removal Agent (MRA) was developed to combat the widespread problems caused by contamination of cell cultures with mycoplasma.
Very effective at removing mycoplasma from infected cell cultures.
Shows strong anti-mycoplasma activity against many types of mycoplasma includingMycoplasma orale, M. arginini, M. hyorhinis andAcholeplasma laidlawii.
Suitable for use to prevent recontamination of the culture with the original mycoplasma.
Can be used to prevent initial infection of cells in culture by mycoplasma.
Non toxic — will not interfere with the viability or function of cells in culture.
Very easy to use — simply incubate for a week after addition to mycoplasma-contaminated cell cultures.
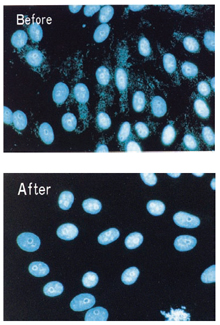
The effect of Mycoplasma Removal Agent (0.5 mg/ml) in the culture media of mycoplasma-infected cells after 1 week.
Add MRA to cell cultures contaminated by mycoplasma at a concentration of 0.5 μg/ml and incubate for a week.
For media replacement or culture transfer (passage), use a medium containing MRA at this same concentration.
Transfer the cell cultures several times without MRA and confirm that regrowth of the contaminating mycoplasma has not occurred.
If mycoplasma in serum or trypsin cannot be ruled out, MRA can be added to the media at a concentration of 0.5 μg/ml to prevent contamination of cell cultures exposed to these products.
Note: The recommended MRA concentration for use is 0.5 μg/ml. The MRA concentration should be raised up to 1 μg/ml only if the recommended concentration is ineffective in removing the mycoplasma.
Cytotoxicity of MRA is low and cell toxicity is rare when used at the recommended concentration. However, it is recommended that the retention of desired cellular characteristics be confirmed after treatment.
Note: Mycoplasma Removal Agent should not be used as a substitute for good cell culture techniques!
Nakai, N. et al. (2000). Detection and elimination of contaminating microorganisms in Transplantable Tumors and Cell Lines. Exp. Anim. 49: 309 – 313.
MRA is stable at room temperature. Protect from light to prevent decomposition.
5 years from date of despatch.